Authors: Alec Edwards
Lisa Rothmann
Universtiy of the Free State UFS
Department of Plant Science, Plant Pathology
Introduction
Sclerotinia sclerotiorum is one of the most devastating soilborne plant pathogens and affects more than 500 plant species, including important oilseed and protein crops. This pathogen has become a significant threat to production of crops such as sunflower and soybean in South Africa. The primary sign of Sclerotinia disease is white cottony mycelium with subsequent production of melanised hyphal masses, survival structures called sclerotia.
What complicates disease management?
One of the primary reasons why control of Sclerotinia diseases are so difficult is related to the biology of the pathogen and its complex interactions with the host crop and environment. Sclerotia can persist on plant debris or in the soil for extended periods of time. Eliminating these sources of inoculum from fields remains a challenge. Sclerotia can lead to the infection of plants by means of ascospores released from apothecia and by mycelial infection through the soil, both germinating from sclerotia under differential conditions. Recent research indicates that there is great potential in utilising microorganisms, plant extracts and resistance inducers (RI’s) to control plant diseases.
Current control of Sclerotinia diseases relies heavily on cultural practices and fungicides. Cultural control aims to reduce the number of sclerotia in the soil or to create conditions that are unfavourable for disease initiation. Examples of these include crop rotations and lowering plant density. Limited fungicides are registered for control of Sclerotinia diseases in South Africa. Benomyl is used for sunflower, whereas Procymidone is registered for soybean. The cost of buying, applying fungicides and the potential requirement for multiple applications are economic risks for producers. Fungicide efficiency is highly dependent on the ability to predict the infection establishment in the crop and correctly timing applications. Failure to correctly time applications can result in substantial losses to the producer. Furthermore, the irresponsible use of fungicides pose risks to the environment and human health.
Alternative control, through biological control agents
Due to the risks and complexity surrounding chemical control, researchers investigated alternative methods for managing this pathogen. The use of biological control agents (BCA’s) that directly parasitise the pathogen have shown great potential. Researchers have also been focusing on utilising RI’s to activate the inherent defence mechanisms of the host plant to reduce disease severity.
Fungal BCA's
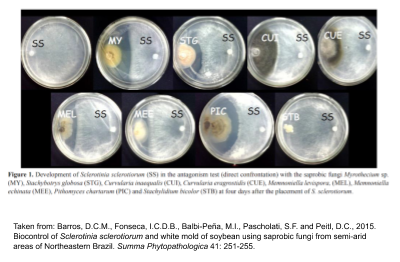
Trichoderma species are hyperparasitic against S. sclerotiorum Figure 1. Responsible for directly parasitising mycelial growth, inhibiting carpogenic and ascospore germination as well as reducing apothecia density under laboratory conditions. Moreover, by-products produced by the fungi not only reduce disease incidence but also promote seedling emergence and increase soybean grain yield under field conditions. However, different strains within species of the Trichoderma genus result in varying degrees of disease control.
Intense field and glasshouse studies have been conducted with the parasitic fungus Coniothyrium minitans which is part of commercial formulations. Damage caused by S. sclerotiorum is reduced by infecting and degrading sclerotia in the soil. C. minitans is most effective when applied to the soil prior to planting and key to its success is its ability to persist and spread within the soil. However, some S. sclerotiorum strains reported produce varying amounts of oxalate (a determining factor of the pathogens virulence) and respond differentially to C. minitans. This mycoparasite remains one of the most promising control options for destroying field-borne sclerotia.
Bacterial BCA's
A two year field study indicated that Pseudomonas cholororaphis and Bacillus amyloliquefaciens can be used to control Sclerotinia stem rot of canola, results were comparable to Iprodione, an active ingredient of fungicides. In 2015, the sclerotia-inhabiting strain B. cereus SC-1, demonstrated potential in reducing the incidence of canola stem rot under controlled and field conditions. Bacillus species that indicated antagonistic activity against S. sclerotiorum also showed plant growth promoting properties. However, Bacillus strains only had significant impacts on disease reduction and growth promotion when they were applied in combination. The majority of characterized bacterial mycoparasites target ascospores and the growing hyphae of S. sclerotiorum. Typically, broth cultures and/or cell suspensions of the bacterial BCA’s are applied to the crop’s aboveground organs.
Although the levels of control achieved in some of the above mentioned experiments are comparable to that of synthetic fungicides, contradicting evidence does exist. Cognisance must be taken of the research published on BCA’s that do not include field trials and or show inconsistent results under field conditions. Thus conclusions made about the efficacy of BCA’s cannot always be accepted as the expected result under field conditions. Environmental factors influence the interaction of BCA’s with the host and the pathogen, thus further field research and product development is needed to advance the effectiveness of BCA’s within sustainable cropping systems. However, this does not take away from the inherent abilities of BCA’s to inhibit and even parasitize this pathogen.
Resistance inducers
As a result of the agricultural industry seeking sustainable crop production methods and relying less on chemical control a recent interest in the use of resistance inducers (RI’s) has been observed. A wide range of RI’s exist, their primary effect is to trigger the host crop’s internal defence mechanisms. The mode of action of RI’s are diverse but work on the same premise,activating the systemic acquired resistance (SAR) in plants. SAR leads to the initiation of different defence mechanisms ranging from reinforcing cell walls to increasing defence enzymes. The activation of SAR in plants leads to resistance against a broad range of pathogens. A well-known chemical inducer of plant defence include plant hormone is salicylic acid (SA) and its derivatives. Salicylic acid is effective at very low concentrations and induces plant growth promotion, yield and increases microbial activity in the rhizosphere, the micro-environment surrounding the root system. SAR to S. sclerotiorum has been induced in mature kiwifruit vines with SA applications and a pre-treatment of SA + SA-derivative reducing subsequent lesion size. This serves as a promising tactic to manage damage incurred by Sclerotinia diseases on high value crops.
Chitosan is an abundant natural polymer with nontoxic, biodegradable and biocompatible properties. Chitosan has dual effects, controlling the ability of microorganisms to sporulate, germinate and infect it also induces defence responses in the host plant. Chitosan is known to enrich rhizosphere biodiversity. A richer rhizosphere diversity can lead to a disease suppressive environment where microorganisms can outcompete S. sclerotiorum. Low concentrations of chitosan and SA, have been shown to inhibit mycelial growth of S. sclerotiorum in the laboratory. Inducing resistance leads to proactive protection and can also lead to positive crop growth, yielding healthier plants.
Conclusion
A plethora of evidence suggests BCA’s and RI’s have the potential to control Sclerotinia diseases. As with all disease management strategies, they too have limitations, requiring favourable environmental conditions to establish and be effective. They also have to be applied prior to infection and sometimes as close to the event as possible, thus monitoring the environmental conditions is key to the success of disease management. Ultimately, the use of biological control requires an integrated approach, with a commitment to the long-term establishment within a cropping system.
References
Please contact primary author: EdwardsA@ufs.ac.za